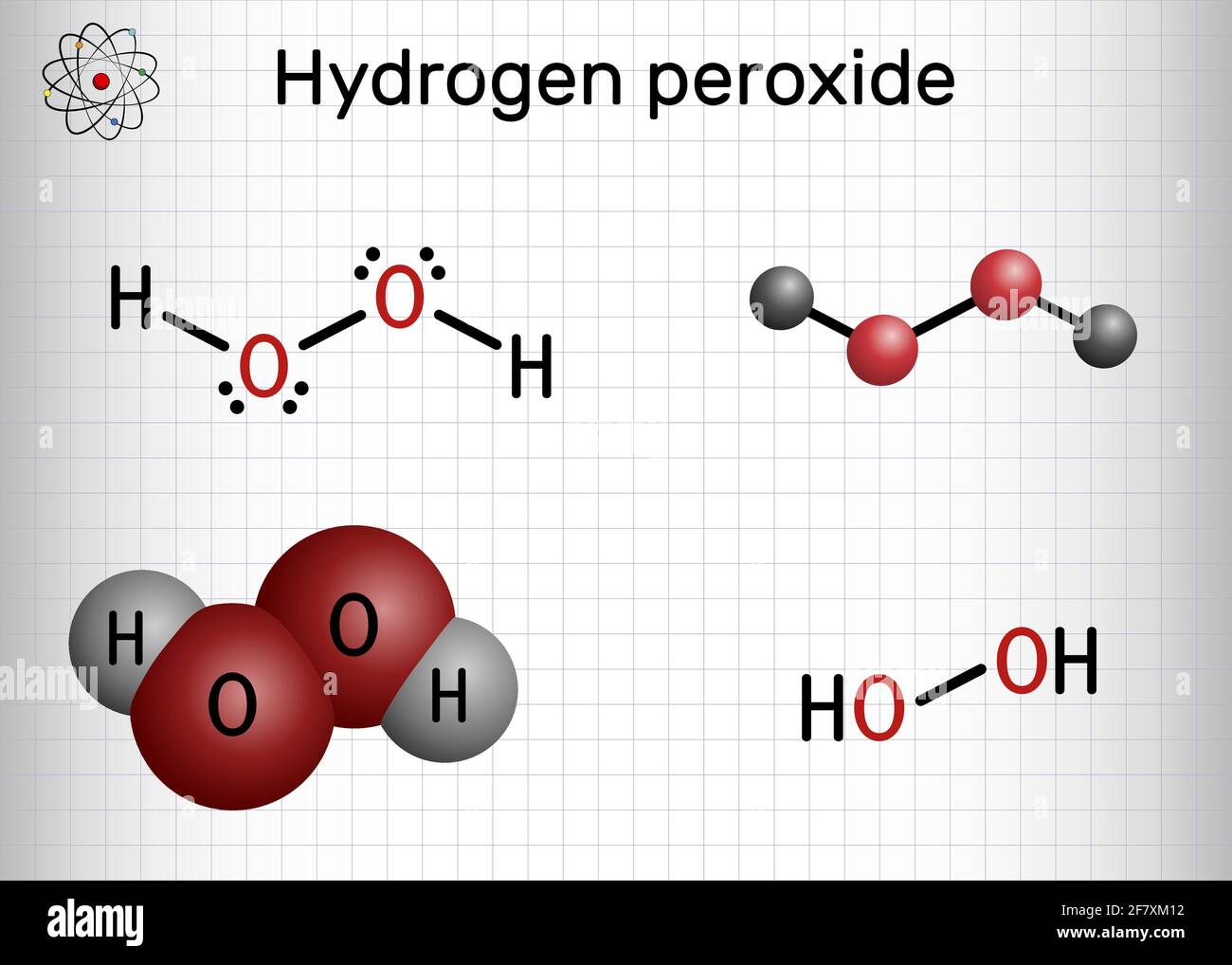
Hydrogen peroxide (H2O2) can act as oxidising as well as reducing agent in both acidic and alkaline media. From the following given reactions, select the option(s) in which reducing action of H2O2

Comment équilibrer : H2O2 → O2 + H2O (peroxyde d'hydrogène, dioxygène, eau) | Physique-Chimie - YouTube

SOLVED: Below is a proposed mechanism for the decomposition of H2O2. H2O2 + I- â†' H2O + IO- (slow) H2O2 + IO- â†' H2O + O2 + I- (fast) Which of the

2H202 alkaline medium *2H20 + 02 the proposed mechanism is as given below : (1) H2O2 +1 → H2O+IO (slow) (2) H202 + 10 + H20+1+02 (fast) (i) Write rate law the
I) H2O2 + O3 → H2O + 2O2 (II) H2O2 + Ag2O → 2Ag + H2O + O2 Role of hydrogen peroxide in the - Sarthaks eConnect | Largest Online Education Community

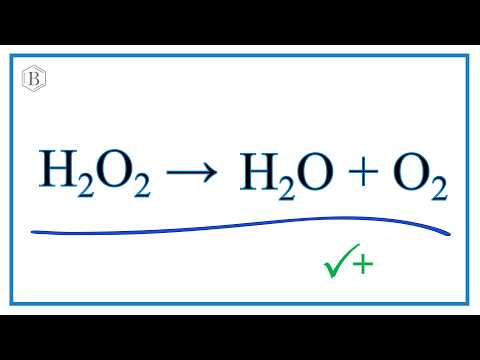
H2O2=H2O+O2 balance the chemical equation @mydocumentary838. h2o2=h2o+o2 balance the equation. - YouTube

Clicker #1 Which of the following shows the correctly balanced equation for this reaction: H2O2 → H2O + O2 A) 3 H2O2 → 3 H2O + O2 B) H2O2 → 2 H2O + O2. - ppt download

H2O2=H2O+O2 balance the chemical equation @mydocumentary838. h2o2=h2o+o2 balance the equation. - YouTube