Anémomètre sonique 3D et analyseur rapide CO2/H2O placés sur une "tour à flux" et mesurant les échan | CNRS Images
Write fully balanced equations for the following : (a) CO2 + H2O → ............ - Sarthaks eConnect | Largest Online Education Community

of breakthrough experiments and deduced competitive CO2–H2O loadings... | Download Scientific Diagram

Figure 1 from The CO2-H2O system: III. A new experimental method for determining liquid-vapor equilibria at high subcritical temperatures | Semantic Scholar
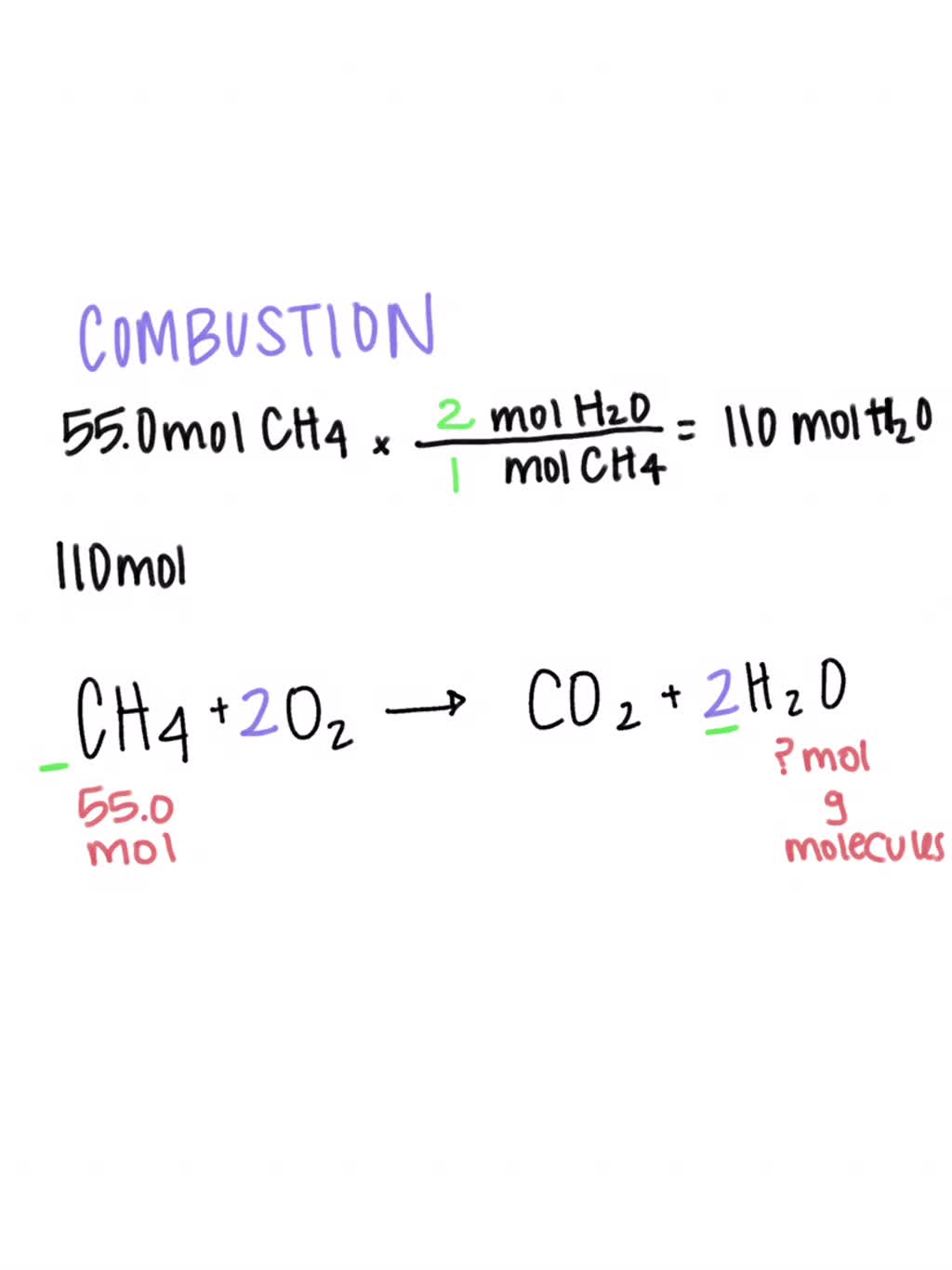
SOLVED: CH4 + O2 ——————- CO2 + H2O What type of reaction does this equation represent? Write and balance the equation. Assume that there are 55.0 moles of CH4. How many moles